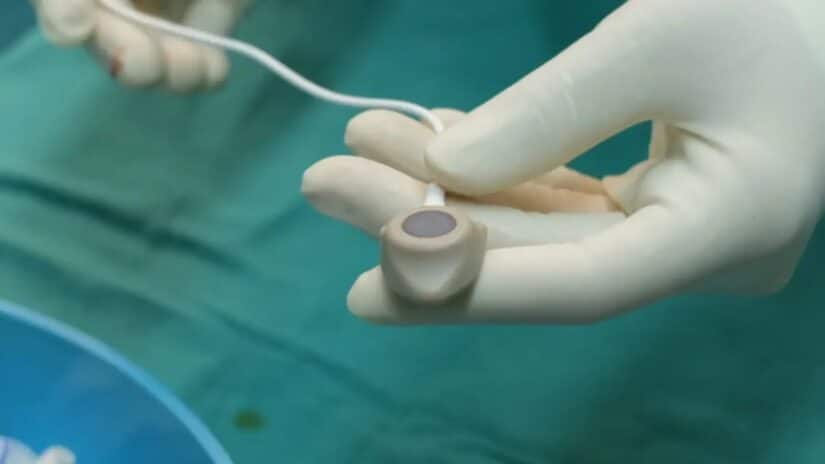
The Bard PowerPort, a widely used port catheter device designed for chemotherapy and intravenous delivery has come under intense scrutiny after thousands of patients reported serious medical complications. These patients have filed PowerPort Lawsuits claiming the device was defectively designed and caused severe injuries such as blood clots, organ damage, and catheter fractures.
As of September 2025, more than 1,900 Bard PowerPort Lawsuit cases have been consolidated into a multidistrict litigation (MDL No. 3081) in the U.S. District Court. The litigation alleges that Becton Dickinson, the parent company of Bard Access Systems Inc., failed to properly test and warn patients about the risks of the PowerPort device, leading to widespread injuries and even wrongful death. Individuals affected by these complications may benefit from speaking with a product liability lawyer to understand their rights and explore potential options for compensation.
What Is the Bard PowerPort and Why Lawsuits Are Rising
The Bard PowerPort is a port catheter placed under the skin. Doctors use it for chemotherapy and long-term infusions. It was marketed as safe and effective. However, many patients reported serious issues.
These issues include device breakage, catheter movement, and blood clots. As a result, lawsuits have increased across the U.S.
Bard Access Systems, a unit of Becton Dickinson, made this device. It uses a flexible material called Chronoflex AL. However, many claims say this material weakens over time. Therefore, it may fail inside the body.
Common Complications Linked to the Device
Patients have reported several serious problems. First, blood clots can form and travel to the lungs. This can lead to stroke or organ failure.
Second, the device may break apart. Broken pieces can move through the bloodstream and damage organs.
Third, the port may shift inside the body. This can cause drug leaks and tissue damage.
In addition, infections may occur. Bacteria can enter the bloodstream and cause sepsis.
Because of these risks, many patients need emergency surgery to remove the device.
FDA Warnings and Recalls
The FDA has tracked complaints about this device for years. In 2020, it announced a recall due to breakage risks.
Later recalls also involved similar devices. For example, some had sterility issues. Others had weak catheter tubing.
Many experts criticize the approval process. They say it allowed unsafe devices into the market. Therefore, the FDA continues to review reports and medical data.
The Growing PowerPort Lawsuit
Many cases are now part of a federal multidistrict litigation (MDL). This process groups similar lawsuits together.
Judge David G. Campbell oversees the case. Plaintiffs share evidence and expert reports.
They claim the company sold a defective product. They also say it failed to warn about serious risks.
Bellwether Trials in 2026
The court has scheduled several test trials, known as bellwether trials. These trials will guide future outcomes.
The first trial starts in March 2026. Additional trials will follow throughout the year.
These results may lead to settlements or further legal action.
Key Allegations Against the Company
Plaintiffs claim the company made several mistakes. First, it sold a defective device.
Second, it ignored known risks. Third, it failed to warn doctors and patients.
They also argue the company prioritized profit over safety. Therefore, many victims are now seeking compensation.
Role of Becton Dickinson
Becton Dickinson acquired Bard in 2017. After that, reports of device issues continued.
Lawsuits claim the company failed to fix known problems. Some internal records suggest early awareness of defects.
As a result, the company now faces increased legal pressure.
Impact on Patients and Families
Many patients face serious health issues after using this device. They often need surgery and long-term care.
Medical costs and missed work create financial stress. In severe cases, families have filed wrongful death claims.
Therefore, the impact goes beyond physical injury.
What Victims Should Do
Patients should document all medical issues and treatments. They should also keep hospital records and reports.
Next, they should consult a lawyer experienced in medical device cases. This helps them understand their rights.
Victims may claim compensation for medical bills, lost income, and pain. In some cases, families may file wrongful death claims.
Taking action early can improve the chances of a strong case.
About Ted Law
At Ted Law Firm, We serve families across Aiken, Anderson, Charleston, Columbia, Greenville, Myrtle Beach, North Augusta and Orangeburg. Our goal is to educate and empower patients impacted by medical complications, manufacturing defects, and design defects caused by unsafe medical devices.Contact us today for a free consultation